| 中文名称: | Stampidine | ||||
|---|---|---|---|---|---|
| 英文名称: | Stampidine | ||||
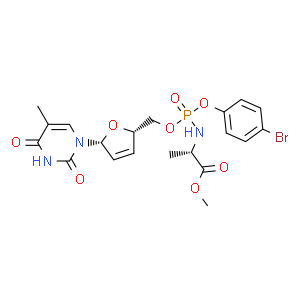
| 别名: | Stampidine methyl (2R)-2-[[(4-bromophenoxy)-[[(2S,5R)-5-(5-methyl-2,4-dioxopyrimidin-1-yl)-2,5-dihydrofuran-2-yl]methoxy]phosphoryl]amino]propanoate | ||||
| CAS No: | 217178-62-6 | 分子式: | C20H23BrN3O8P | 分子量: | 544.29 |
| CAS No: | 217178-62-6 | ||||
| 分子式: | C20H23BrN3O8P | ||||
| 分子量: | 544.29 | ||||
基本信息
|
产品编号:S10645 |
|||||
|
产品名称:Stampidine |
|||||
|
CAS: |
217178-62-6 |
储存条件 |
粉末 |
-20℃ |
四年 |
|
分子式: |
|||||
|
分子量 |
544.29 |
||||
|
化学名: |
|||||
|
Solubility (25°C) |
体外 |
DMSO |
|
||
|
Ethanol |
|
||||
|
Water |
|
||||
|
体内 |
现配现用 |
|
|||
|
<1mg/ml表示微溶或不溶。 |
|||||
|
普西唐提供的所有化合物浓度为内部测试所得,实际溶液度可能与公布值有所偏差,属于正常的批间细微差异现象。 |
|||||
|
请根据产品在不同溶剂中的溶解度选择合适的溶剂配制储备液;⼀旦配成溶液,请分装保存,避免反复冻融造成的产品失效。 |
|||||
生物活性
|
产品描述 |
一种核苷逆转录酶抑制剂 (NRTI),具有高效、广谱的抗 HIV 活性。 |
||||
|
靶点/IC50 |
HTLVIIIB |
primary clinical isolates |
NRTI-resistant primary clinical isolates |
NRTI-resistant primary clinical isolates |
|
|
IC50:1nM |
2nM |
8.7nM |
11.2nM |
||
|
体外研究 |
Stampidine (7.8-1,000μM; 24 hours) is not cytotoxic to genital tract epithelial cells.Stampidine has no effect on sperm motility in cervical mucus. |
||||
|
体内研究 |
Stampidine (50-100mg/kg; p.o.) exhibits potent antiretroviral activity in chronically feline immunodeficiency virus (FIV)-infected cats. Stampidine (100mg/kg; p.o.) shows the average plasma Cmax, AUC, half-life (t1/2), and mean residence time (MRT) values of15.4 µM, 23.1 µM•h, 108.6 min and 119.4 min, respectively, in dogs. Stampidine does not cause anemia, thrombocytopenia, neutropenia, or lymphopenia suggestive of hematologic toxicity,elevations of BUN or creatinine or electrolyte disturbances suggestive of renal toxicity or metabolic abnormalities,elevations of ALT, AST, Alk in adult beagle dogs. |
||||
|
Animal Model: |
SPF male or female domestic cats (2.9- 6.2kg), with chronically FIV-infected |
||||
|
Dosage: |
50mg/kg, 100mg/kg |
||||
|
Administration: |
Oral administration (oral bolus dose) |
||||
|
Result: |
Oral administration (oral bolus dose). |
||||
|
Animal Model: |
Male beagle dogs (10-12kg) |
||||
|
Dosage: |
100 mg/kg (Pharmacokinetic Analysis) |
||||
|
Administration: |
Oral administration |
||||
|
Oral administration |
The estimated average plasma Cmax and AUC values were 15.4 µM and 23.1 µM·h, respectively. The average elimination half-life (t1/2) and mean residence time (MRT) were 108.6 min and 119.4 min, respectively. |
||||
本计算器可帮助您计算出特定溶液中溶质的质量、溶液浓度和体积之间的关系,公式为:
质量 (g) = 浓度 (mol/L) x 体积 (L) x 分子量 (g/mol)
摩尔浓度计算公式
用本工具协助配置特定浓度的溶液,使用的计算公式为:
开始浓度 x 开始体积 = 最终浓度 x 最终体积
稀释公式
稀释公式一般简略地表示为:C1V1 = C2V2 ( 输入 输出 )